[ad_1]
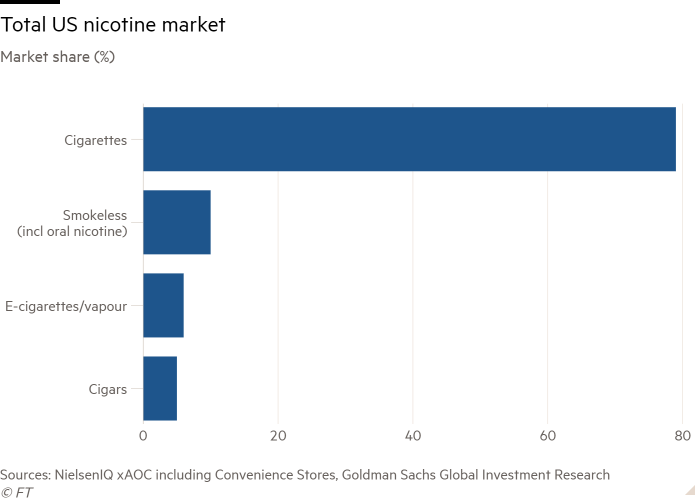
The authorisation of the primary e-cigarette by US regulators this week was described by business consultants as a historic second for the $5bn sector, which has been working underneath a cloud since a crackdown on a teen vaping “epidemic” in 2018.
However the US Meals and Drug Administration’s alternative of product for its first ever inexperienced mild — an unpopular gadget formed like an actual cigarette made by a subsidiary of British American Tobacco — puzzled some observers.
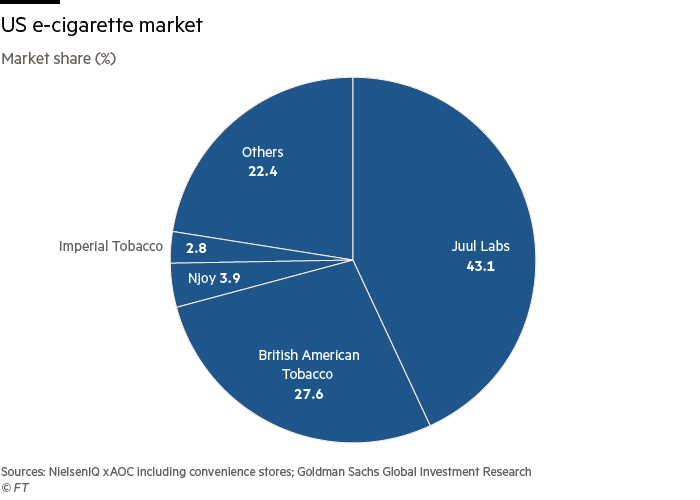
Juul, the market chief with a 43 per cent share, has additionally filed for authorisation, and has but to seek out out whether or not its merchandise can be authorised too.
Nonetheless, by rubber stamping the Vuse Solo e-cigarette and associated units, the company despatched a transparent sign that it doesn’t intend to ban vaping outright. Fairly, it began to sketch out a brand new regulatory regime the place it would enable merchandise designed to assist folks give up smoking whereas clamping down on these aimed toward first time vapers who are usually not hooked on tobacco — particularly youngsters.
“It’s a historic second and an inflection level within the lengthy, drawn out and controversial means of tobacco laws,” stated Cliff Douglas, director of the College of Michigan Tobacco Analysis Community.
Simply as essential was the company’s choice to reject 5 flavoured tobacco merchandise additionally made by Reynolds American, the BAT subsidiary that markets the Vuse Solo. In doing so, the FDA expressed its distaste for the sorts of vaping merchandise that had led to an explosion in utilization amongst teenagers.
It has additionally declined, to date, to rule on BAT’s rather more common gadget, the Vuse Alto, and positioned strict advertising curbs on the Solo product.
“The FDA is underneath huge political stress and is making an attempt to string a needle in a method that satisfies its scientific and public well being mission within the face of litigation, threats and political hardball,” stated Douglas.
He stated the denial of flavours by the FDA clearly signalled it will take a conservative method to any merchandise that have been common amongst college kids and younger folks.
Final 12 months, US regulators briefly banned the manufacture, distribution and sale of cartridge-based fruit and mint flavoured e-cigarettes to sort out what they known as an “epidemic” of teenage vaping. The FDA required all US e-cigarette producers to submit their merchandise for overview or take them off the market.
Even earlier than the FDA formalised its crackdown, the rising regulatory uncertainty had sparked a administration shake-up at Juul in 2019 and prompted Philip Morris Worldwide to name off talks a few $200bn merger with Altria, which owns a 35 per cent stake in Juul.
Juul has since reduce on advertising spending and stopped promoting flavoured pods for its vaporiser gadget.
The strategic reset has value the corporate market share, which has fallen from 64 per cent in Might 2018 to 43 per cent final month. Retail gross sales of Juul merchandise fell 10 per cent to $1.99bn within the 52 weeks to September 25, based on a Goldman Sachs evaluation of Nielsen information.
However Juul is assured that by repositioning itself as a accountable business chief centered on grownup people who smoke it could possibly scale back regulatory threat and construct a sustainable enterprise over the long run.
Final month the corporate launched a brand new Juul2 gadget within the UK, which incorporates new expertise to forestall the usage of counterfeit pods and make its units much less accessible to kids.
“Over the previous a number of years, we reset our firm as a result of whereas tens of millions of grownup people who smoke have transformed to our merchandise from cigarettes, we are going to solely be trusted to offer options to grownup people who smoke if we proceed to fight underage use,” stated Joe Murillo, Juul’s chief regulatory officer.
Reynolds has benefited from Juul’s strategic pivot and has continued advertising aggressively over the previous 12 months. It additionally lower the worth of its primary e-cigarette gadget to 99 cents, one-tenth of what Juul’s units usually value. Retail gross sales of its e-cigarette merchandise surged 60 per cent to $1.2bn within the 12 months to September 25, based on Goldman evaluation.
Analysts stated the FDA’s approval of the Vuse Solo product was unlikely to offer Reynolds with a lot of a aggressive benefit as a result of it’s such a small a part of its e-cigarette gross sales — about 1.5 per cent.
“The FDA is authorising a tiny, mainstream vapour product in Vuse Solo — a product not even featured on the Vuse model web site,” stated Christopher Growe, analyst at Stifel, an funding financial institution.
He stated the FDA’s choice was maybe extra noteworthy for what it had not authorised: Reynolds’ primary e-cigarette product Vuse Alto, the a lot slicker e-cigarette which accounts for the overwhelming majority of the corporate’s market share.
Reynolds stated it was assured within the high quality of its utility to the FDA for Alto, which was submitted practically a 12 months after the applying for Solo.
However business and well being consultants say the FDA’s choice to reject the corporate’s utility for flavours and severely limit advertising for Solo despatched a robust message to the business about future approvals.
“It was unclear till now if the FDA would take the same method to the bigger and extra highly effective corporations,” stated Desmond Jenson, a lawyer specialising in tobacco on the Mitchell Hamline College of Legislation. “Now they’ve . . . That half is excellent information for public well being.”
However the FDA nonetheless has large selections to make on whether or not to authorise menthol e-cigarette merchandise, one of the crucial common classes, and Juul’s functions, stated Jenson.
The American Vaping Affiliation stated it welcomed the authorisation of the Vuse Solo product however instructed the Monetary Instances it will encourage Reynolds to sue the FDA over its rejection of flavoured merchandise.
Gregory Conley, AVA president, stated banning common flavours would push folks to in direction of gray market merchandise or again to smoking.
At the same time as regulation is catching up with the e-cigarette business new rivals are rising which have to date managed to keep away from FDA scrutiny. Puff Bar, an organization run by vaping entrepreneurs Patrick Beltran and Nick Minas, is promoting merchandise utilizing artificial nicotine — a substance that’s not at present regulated by the FDA.
Critics allege the corporate markets a bunch of flavours akin to banana, watermelon and mint that are common amongst kids. It has grow to be the most important vendor of disposable e-cigarettes, producing $155m in gross sales within the 12 months to September 25, based on Goldman Sachs evaluation.
Puff Bar didn’t instantly reply to a request for remark.
Stifel’s Growe stated Puff Bar was now the primary youth model and the FDA had been caught flat footed once more in being unable to manage the merchandise that have been hottest amongst youthful folks.
“We stay upset within the FDA’s regulatory actions which proceed to punish corporations following its guidelines whereas turning its head on the businesses breaking the principles,” stated Growe.
Further reporting by Andrew Edgecliffe-Johnson in New York
[ad_2]
Source link









