[ad_1]
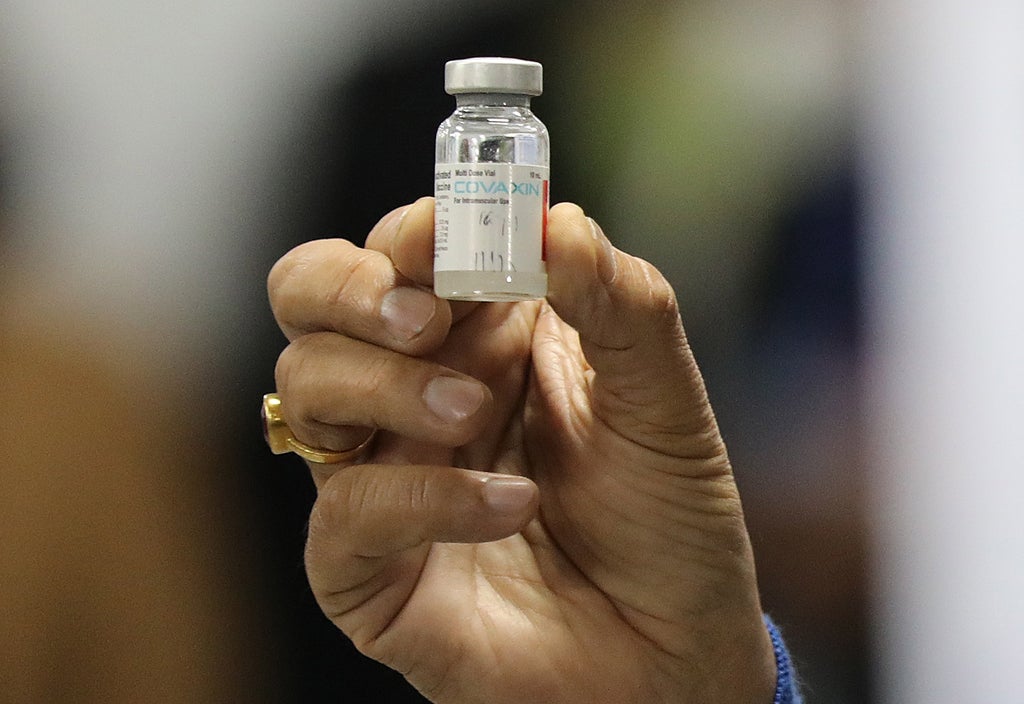
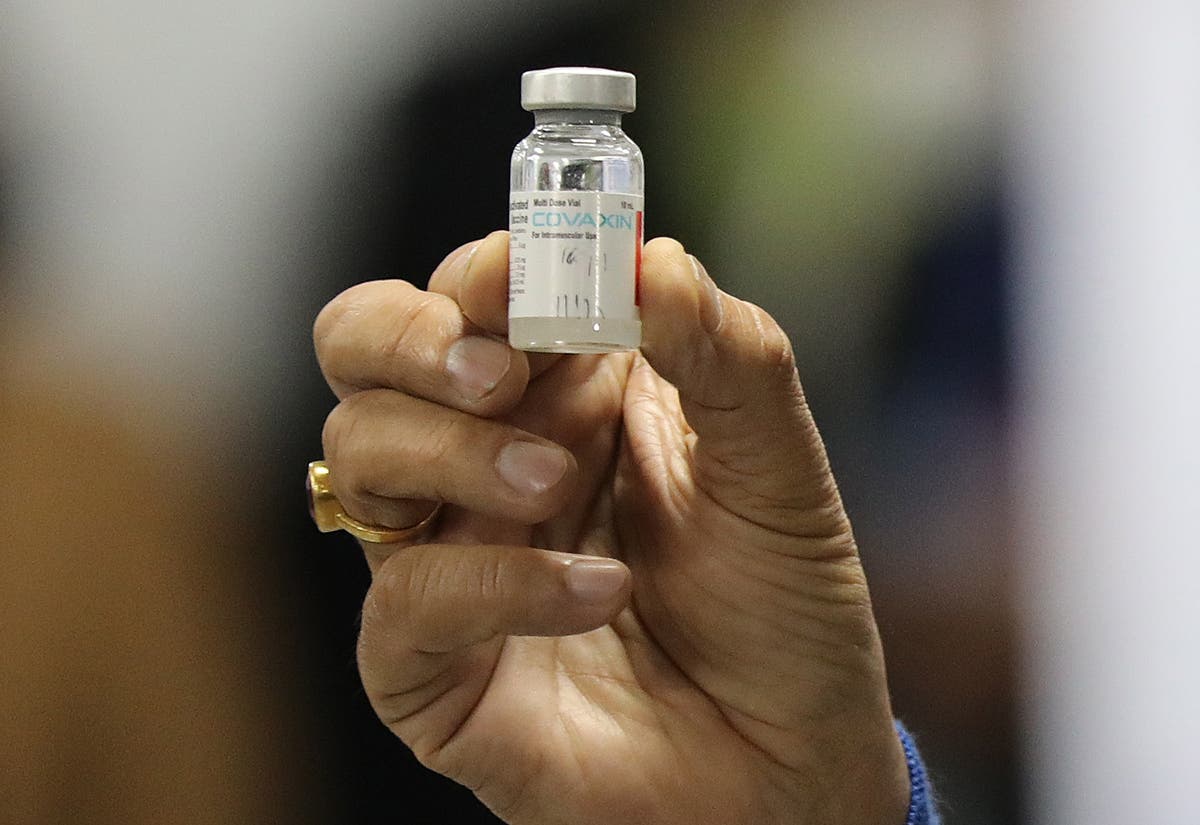
The World Well being Organisation has defended its choice to delay the approval of India’s do-it-yourself coronavirus vaccine Covaxin and mentioned that it can’t lower corners in its course of.
The worldwide well being physique mentioned that it was in search of “one further piece of data” from Bharat Biotech, the Hyderabad-based firm which created the vaccine.
The vaccine, which was India’s first indigenously made Covid jab, is one in every of three at present getting used within the nation’s Covid-19 inoculation drive. India’s medicine management physique had accredited the restricted emergency use of Covaxin in January. It accounts for 11 per cent of the 980 million doses administered within the nation up to now.
“We’re conscious that many individuals are ready for WHO’s suggestion for Covaxin to be included within the #COVID19 Emergency Use Itemizing, however we can’t lower corners — earlier than recommending a product for emergency use, we should consider it totally to verify it’s protected and efficient,” the WHO mentioned in a collection of tweets on Monday.
“Bharat Biotech — the producer of Covaxin — has been submitting knowledge to WHO on a rolling foundation and WHO specialists have reviewed these knowledge. WHO is anticipating one further piece of data from the corporate right now,” it added.
Bharat Biotech first submitted paperwork and the preliminary set of this “rolling knowledge” for WHO approval in July. The well being physique has mentioned it should analyse the knowledge as and when it is available in.
“The timeframe for the WHO Emergency Use Itemizing process depends on how shortly an organization producing the vaccine is ready to present the information required for WHO to judge the vaccine’s high quality, security, efficacy and its suitability for low- and middle-income international locations,” the WHO wrote.
“When the knowledge offered addresses all questions raised, WHO and the Technical Advisory Group will full the evaluation and are available to a last suggestion whether or not to grant Emergency Use Itemizing to the vaccine,” it added.
The WHO’s technical advisory group is scheduled to fulfill on 26 October to think about the itemizing for Covaxin. Nevertheless, the well being physique has not specified which paperwork it has sought from the pharma firm.
The delay in approval for Covaxin has been a priority for Indians who’ve been inoculated with the jab. With out the WHO’s approval, Covaxin is unlikely to be accepted as a legitimate vaccine world wide. Bharat Biotech may also be unable to export its vaccine with out the WHO’s nod.
The approval of Covaxin inside India was itself controversial, because it got here earlier than Bharat Biotech had printed the outcomes of its personal part three trials. The approval was nonetheless largely celebrated as successful story of indigenous manufacturing by Indian prime minister Narendra Modi and his cupboard colleagues.
[ad_2]
Source link